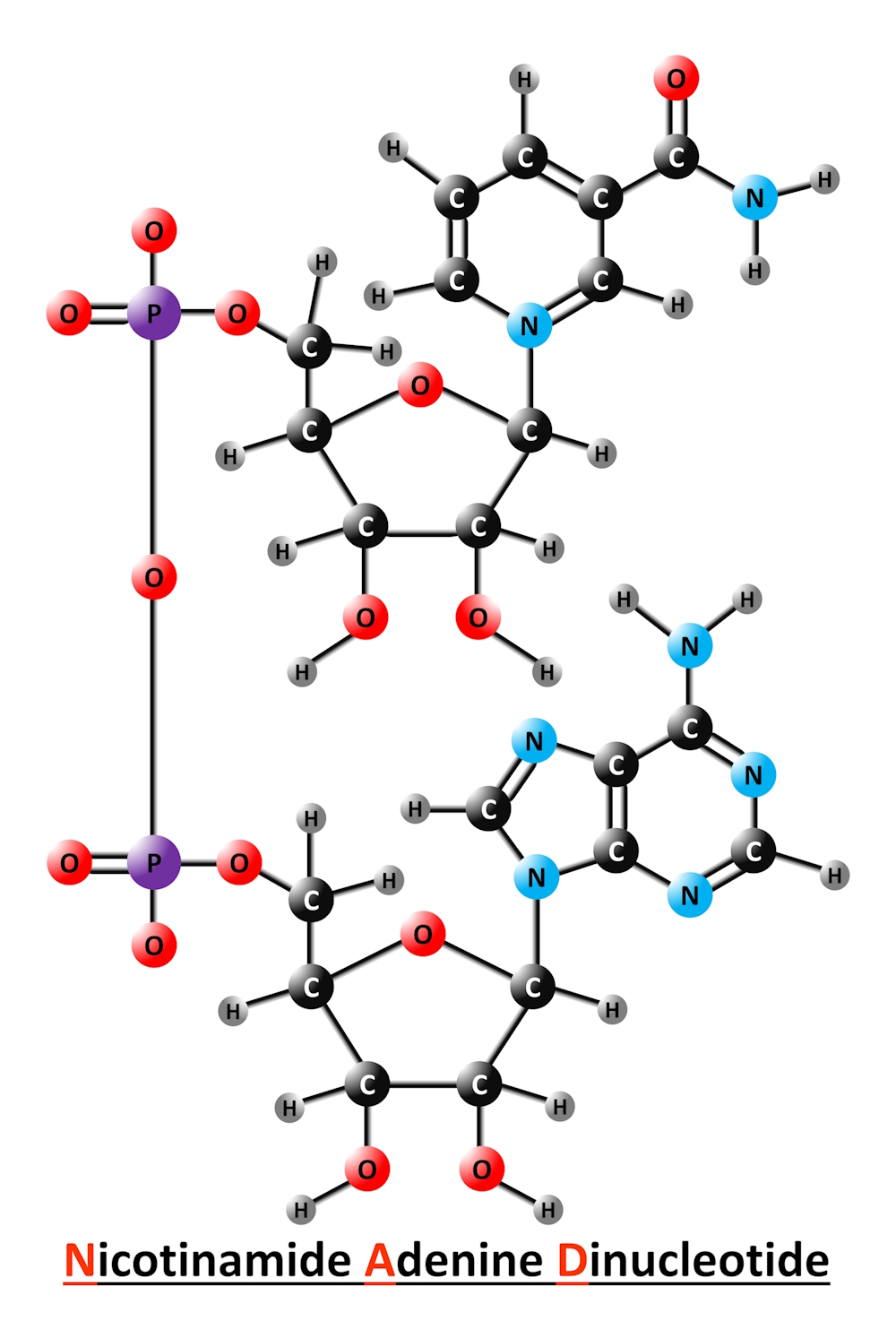
What are NADs?
NADs (nicotinamide adenine dinucleotides) are essential molecules that help signal and convert energy from sugars and fats into a form that muscles and other tissues can use. They exist in four forms: NAD+, NADH, NADP+, and NADPH. The balance of these molecules in different cells and tissues determines how nutrients are used for energy production, growth, and repair.
NAD+ is the oxidized form, vital for energy metabolism and cellular repair.
NADH, the reduced form, is responsible for donating electrons to fuel energy production in mitochondria.
NADP+ is the oxidized form involved in anabolic reactions, such as building molecules.
NADPH, the reduced form, actively safeguards cells from oxidative damage.
NADs regulate and repair living organisms
NADs are necessary to all forms of life – from bacteria and plants to animals and humans – by sustaining biochemical processes in every cell and tissue. In human health, NADs facilitate energy transfer between chemical reactions and support growth and repair functions. These processes drive hundreds of biochemical reactions in our tissues, meaning that any prolonged imbalance in NAD levels can lead to serious health consequences.
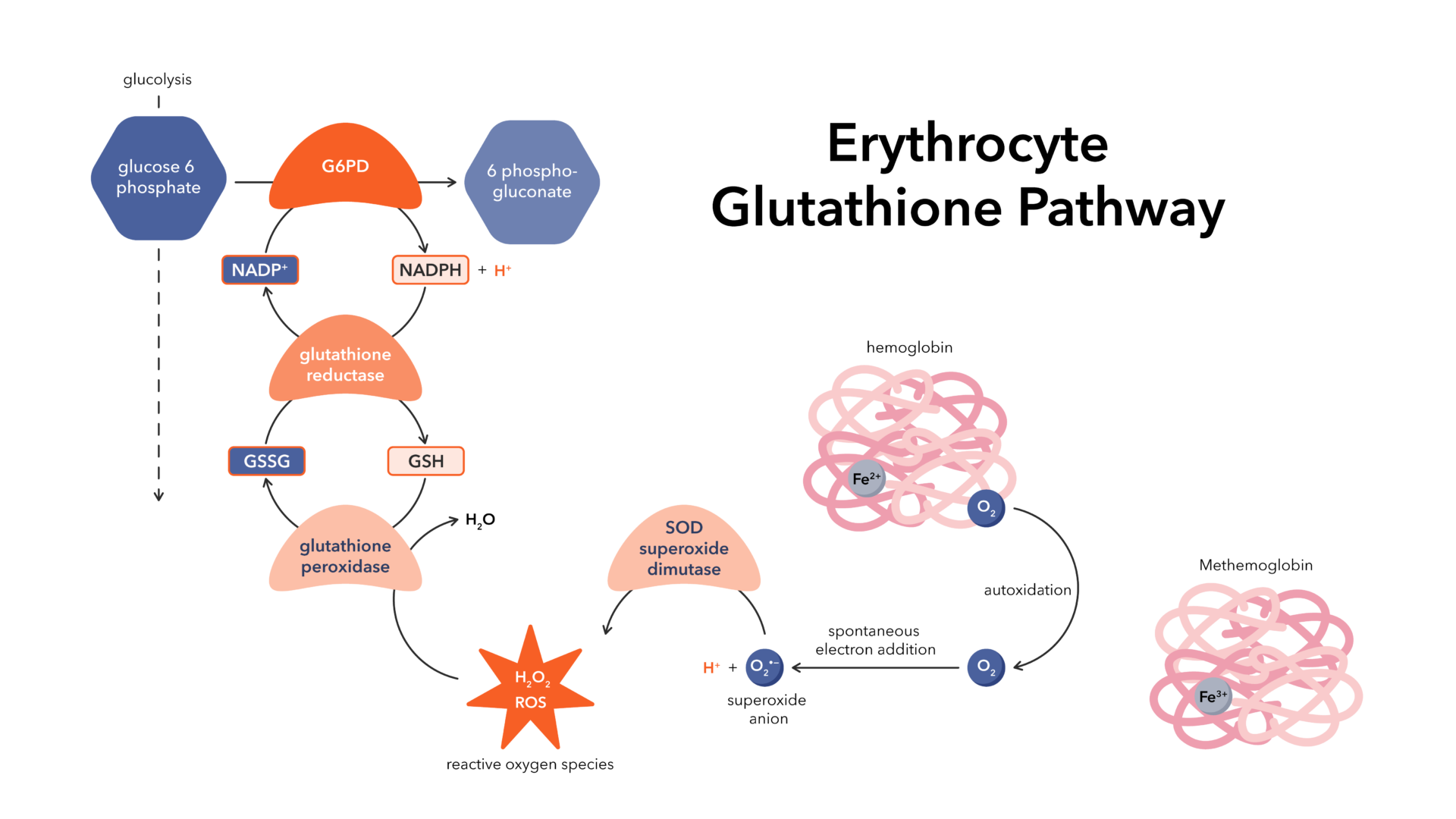
What is glutathione?
Glutathione is a tripeptide made from three amino acids: cysteine, glutamate, and glycine. It exists in two forms: active GSH (reduced) and inactive GSSG (oxidized). Working as a powerful antioxidant, glutathione neutralizes reactive oxygen species (ROS) generated by oxidative stress, which is a natural byproduct of oxygen-dependent metabolism. Beyond its antioxidant role, glutathione plays a vital part in regulating metabolism. Glutathione and NAD are closely connected, as glutathione depends on NADPH to recycle back to its reduced, active form.
NAD and glutathione: Core regulators of cellular redox balance
NAD and glutathione are central to maintaining cellular redox balance, a fundamental property that determines how cells generate energy and respond to stress. NAD exists in oxidized (NAD⁺) and reduced (NADH) forms, acting as a key electron carrier in metabolic reactions. Glutathione, in turn, exists as reduced (GSH) and oxidized (GSSG), forming the primary intracellular antioxidant system.
Together, these two systems provide complementary information. NAD⁺/NADH reflects the state of cellular energy metabolism and electron flow, while GSH/GSSG reflects the oxidative environment and the cell’s ability to neutralize reactive species. Understanding both systems simultaneously allows for a more complete view of cellular physiology, particularly under conditions of metabolic or oxidative stress.
NAD & NADH in cellular energy production
NAD⁺ plays a central role in cellular metabolism by accepting electrons during catabolic reactions such as glycolysis and the tricarboxylic acid (TCA) cycle. In these pathways, nutrients are broken down, and electrons are transferred to NAD⁺, forming NADH. This reduced form then transports electrons to the mitochondrial electron transport chain, where they are used to generate ATP through oxidative phosphorylation.
The balance between NAD⁺ and NADH is critical. A higher NAD⁺/NADH ratio supports efficient metabolic flux and mitochondrial function, while a more reduced state (higher NADH) may indicate altered metabolic conditions. This ratio is therefore not only a reflection of energy production but also a regulator of multiple enzymatic processes linked to cellular homeostasis.
VIDEO: How is NAD produced?
Glutathione system and oxidative stress regulation
Glutathione is the most abundant intracellular antioxidant and plays a key role in detoxifying reactive oxygen species (ROS). In its reduced form (GSH), glutathione donates electrons to neutralize ROS, becoming oxidized (GSSG) in the process. The enzyme glutathione reductase then regenerates GSH from GSSG, maintaining the antioxidant capacity of the cell.
The ratio of GSH to GSSG is widely used as an indicator of oxidative stress. A high GSH/GSSG ratio reflects a reduced, protective environment, whereas a lower ratio indicates increased oxidative pressure. This system is essential for maintaining protein function, membrane integrity, and overall cellular stability, particularly in metabolically active tissues.
NADPH: Connecting metabolism to antioxidant defense
The link between NAD metabolism and the glutathione system is largely mediated by NADPH, a related cofactor generated through pathways such as the pentose phosphate pathway. NADPH provides the reducing power required by glutathione reductase to convert oxidized glutathione (GSSG) back into its active reduced form (GSH).
This connection highlights how cellular metabolism supports antioxidant defense. When metabolic pathways efficiently generate NADPH, the cell can maintain a strong glutathione-based defense system. Conversely, disruptions in NADPH production may compromise the ability to regenerate GSH, linking metabolic imbalance to oxidative stress.
Dynamic interplay under physiological and stress conditions
NAD and glutathione systems are highly dynamic and continuously adapt to changes in cellular conditions. Factors such as inflammation, metabolic demand, environmental stress, and disease can influence both NAD⁺/NADH and GSH/GSSG ratios. These shifts reflect changes in energy utilization, mitochondrial activity, and oxidative burden.
Importantly, these systems do not operate independently. Changes in mitochondrial function can alter reactive oxygen species production, which in turn affects glutathione utilization. At the same time, the availability of NAD⁺ and NADPH influences both energy metabolism and antioxidant regeneration, creating a tightly interconnected network.
Integrated measurement of redox metabolism
Measuring NAD metabolites alongside glutathione provides a more comprehensive assessment of cellular redox status. While NAD⁺ and NADH inform on metabolic activity and mitochondrial electron flow, GSH and GSSG reflect the balance between oxidative stress and antioxidant capacity.
This integrated approach enables a more detailed characterization of cellular state, particularly in contexts where both energy metabolism and oxidative processes are involved. By assessing these complementary systems together, it becomes possible to capture a broader and more nuanced picture of redox biology.
Yes, I want to receive the NADMED Test Interpretation Guide for Practitioners.