Non-destructive redox profiling
across biological matrices
Accurate measurement of redox metabolites is challenging due to their stability requirements upon extraction from a biological matrix. Reduced metabolites such as NADH and NADPH are highly labile and prone to degradation if not stabilized properly, causing artificial shifts in measured levels.
Sample processing can further distort physiological redox balance, particularly in complex biological matrices, as enzymes can catalyse interconversion of NAD forms. In addition, many conventional methods measure only part of the system, limiting the comprehensive interpretation of metabolic state.
NADMED Solution
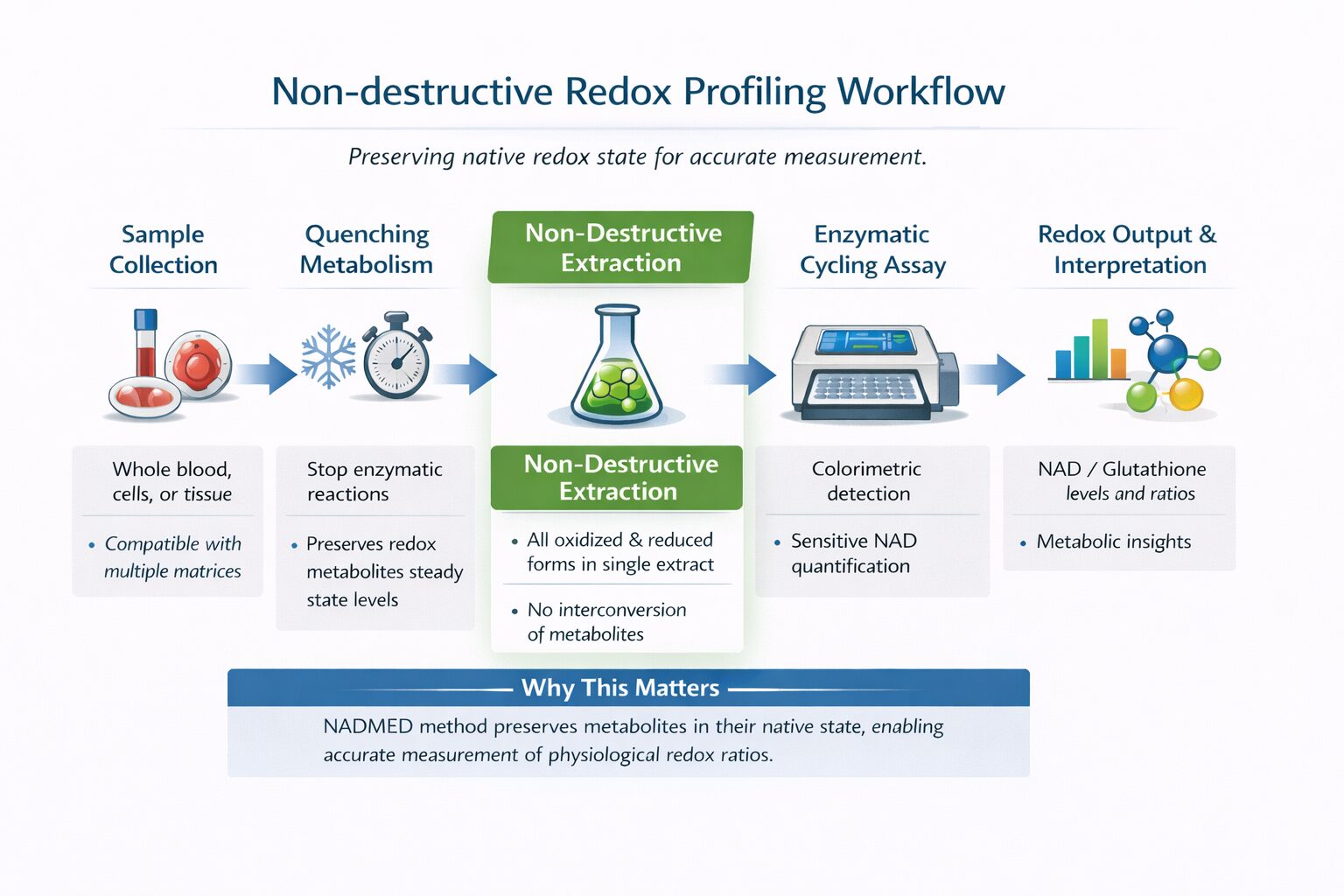
NADMED addresses these challenges through a non-destructive extraction and measurement approach:
Preserves native oxidized and reduced metabolite forms during sample preparation
Minimizes artificial conversion and degradation of redox-sensitive molecules
Enables measurement across whole blood, cells, and tissue
Provides a physiologically relevant snapshot of redox metabolism
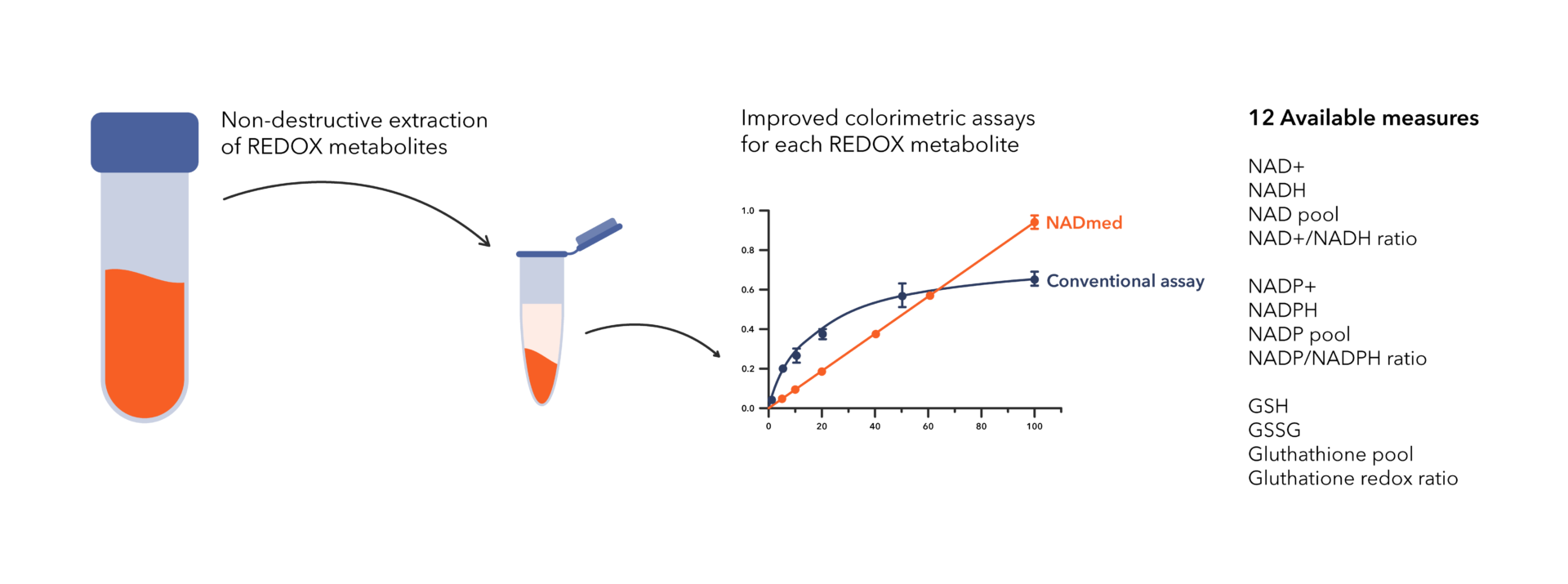
NADMED technology solves two key problems in colorimetric quantification: 1) extraction of metabolites without destroying them and 2) accurate detection of selected metabolites.
Validation and accuracy
The NADMED method is designed to ensure high reproducibility and analytical accuracy, particularly for reduced metabolites that are inherently unstable. Early stabilization combined with non-destructive extraction minimizes artifactual shifts in redox state during sample handling.
This approach enables consistent quantification across different biological matrices and supports reliable comparison between physiological conditions, experimental systems, and study cohorts. It is particularly critical for accurate assessment of redox ratios, which are highly sensitive to pre-analytical variation.

Figure 1:
Repeatability of NADMED test (54 measurements of the same sample; coefficient of variation = 3.94%)
NADMED validation data
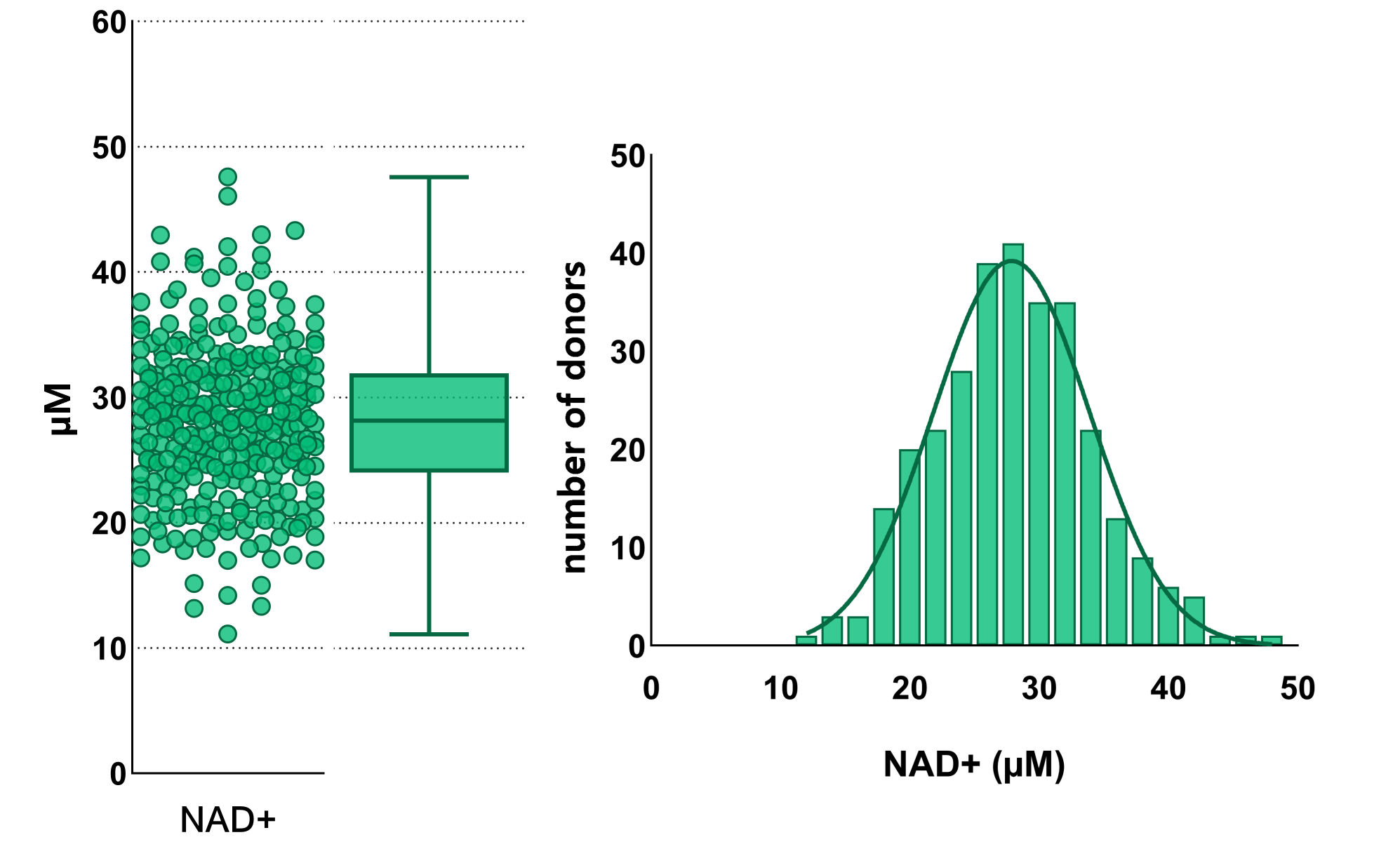
Figure 2:
The largest-ever population dataset of NADs allows for building a reference range, which is required for clinical NAD testing.
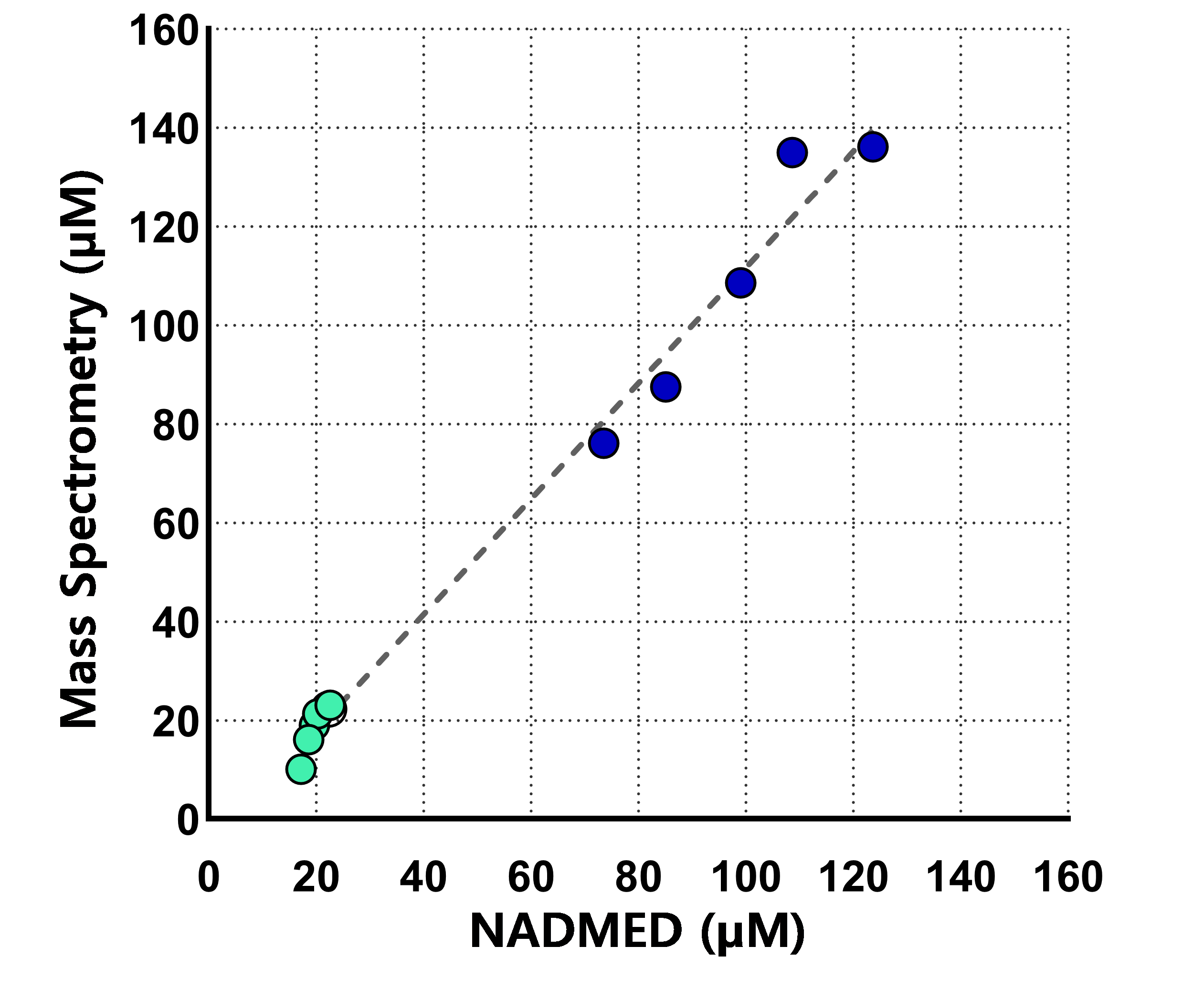
Figure 3:
Correlation of NADMED and mass spectrometry measurement of NAD+ from the same samples.
Key advantages of NADMED method
Complete REDOX profiling of all four NAD and two glutathione metabolites.
CE-marked kits.
Fast and easy: Results for 40 samples in ~4 hours.
No special equipment needed: Works with standard lab readers.
The most accurate test on the market, validated across sample types.
Low sample volume: Only 120 µL of blood or 1,5-2 million cells or 15-20 mg tissue required.
High throughput: Ideal for large-scale studies and clinical use.
Consistent and harmonized results regardless of measurement location.
Biomarker translatability from experiments to clinical trials and clinics.
VIDEO: NADMED CSO Liliya Euro tells about the development of the NADMED method.
How does NADMED method work?
How Does the NADMED Method Work?
Enzymatic cycling enables sensitive, quantitative detection.
The assay is based on enzyme-driven cycling reactions, where NAD metabolites continuously undergo redox conversion, generating an amplified measurable signal. This enables high sensitivity detection in the sub- to low micromolar range, which is critical for extracts of physiological samples.
The method demonstrates strong quantitative performance, with linear response (R² > 0.98) across ~0.3–5 µM, ensuring reliable measurement across biologically relevant concentrations observed in sample extracts.
Preservation of native redox state is critical for accuracy
Redox metabolites are highly sensitive to handling. For example, studies show that >90% of NAD(H) can degrade after freeze–thaw cycle if temperature conditions are not strictly controlled.
The method addresses this by cryopreserving the sample immediately upon collection, using a strictly controlled thawing process developed and validated for blood samples, and applying non-destructive extraction followed by immediate separate stabilization of reduced and oxidized forms. This helps preserve metabolites in their native redox states, which is essential for accurate calculation of redox ratios such as NAD+/NADH, since these ratios are highly sensitive to pre-analytical conditions.
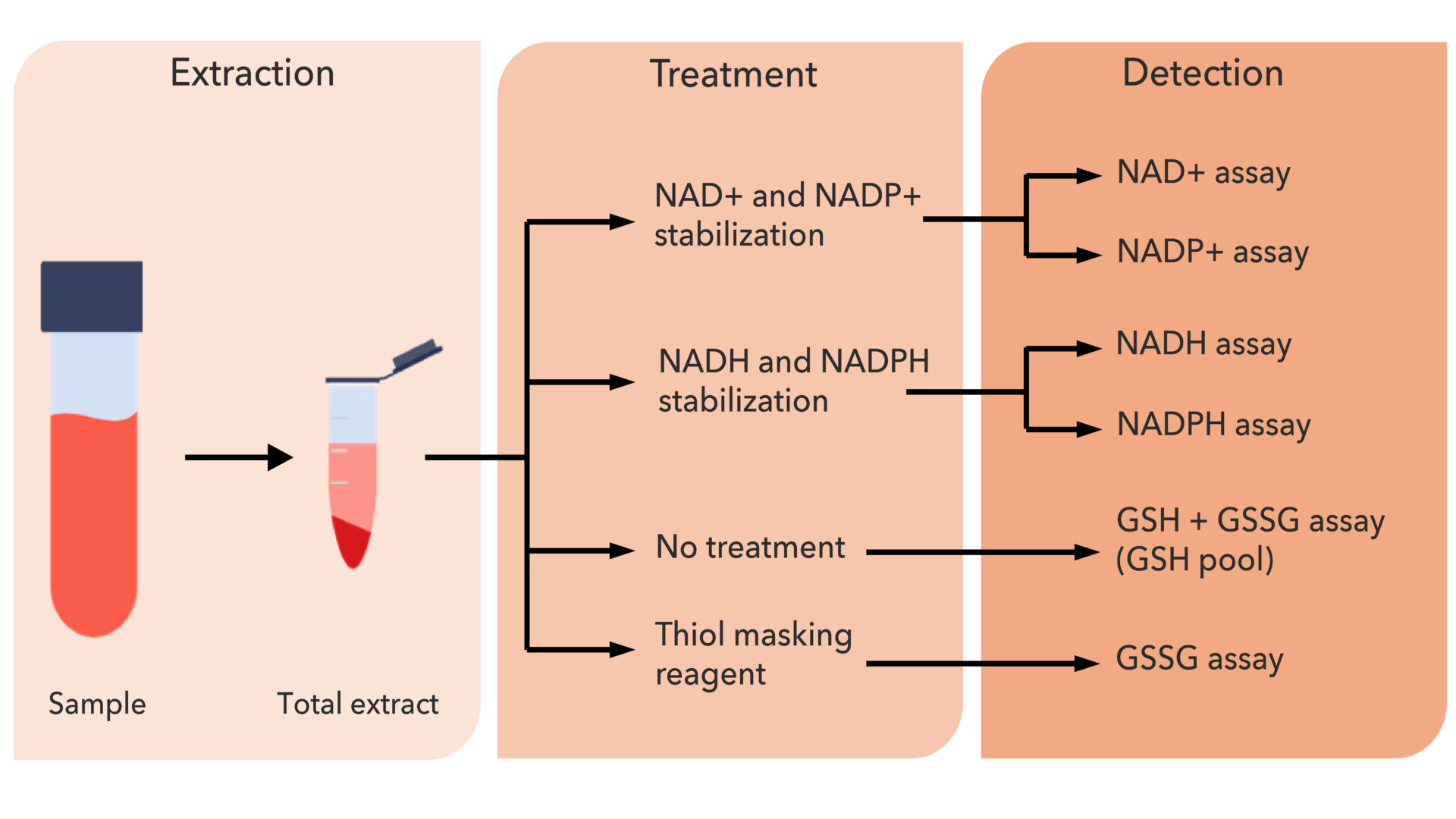
Individual metabolite resolution with high specificity
Each metabolite (NAD⁺, NADH, NADP⁺, and NADPH) is measured individually using selective enzymatic reactions. This avoids pooled signals and enables accurate distinction between closely related redox species.
Reactions in which the target analyte serves as a cofactor or substrate, produce a measurable colorimetric signal that correlates linearly with concentration. Measurements are performed in microplate format using standard spectrophotometric equipment at defined wavelengths (e.g., 565 nm or 600 nm, depending on the analyte).
Broad metabolite coverage across biological matrices
The method enables simultaneous measurement of six key redox metabolites (NAD⁺, NADH, NADP⁺, NADPH, GSH, GSSG) from a single sample, supporting integrated redox analysis.
It has been validated across multiple matrices– including whole blood, cells, and tissue – with whole blood identified as particularly informative due to its ability to reflect systemic metabolic state while minimizing processing steps that can degrade metabolites.
Glutathione redox assessment complements NAD metabolism
Glutathione redox status is assessed through its measurable pool, providing insight into oxidative balance while reducing artefacts from rapid oxidation during handling.
When combined with NAD metabolite data, this enables integrated assessment of cellular redox systems—linking energy metabolism (NAD) with antioxidant capacity (glutathione) for a more complete view of cellular physiology.
Equipment Needed for NADMED Assay
Calibrated Single-Channel Pipettes
Suitable for volume ranges such as:
– 5–50 μL
– 20–200 μL
– 100–1000 μL
Calibrated Multichannel Pipettes
Suitable for volume ranges such as:
– 5–50 μL
– 30–300 μL
Microcentrifuge
Capable of:
– Cooling to +4 °C
– Centrifugal force up to 20,000 × g
Spectrophotometric Microplate Reader
– Must be capable of measuring absorbance at 570–573 nm
– Scanning brightness/intensity must be adjustable
– Set to “low” or adjust to 5–10 flashes per measurement
Dry Bath Heat Block
– Compatible with 1.5 mL microcentrifuge tubes
– Adjustable temperature up to 80 °C